Succession is one of the fundamental concepts in ecology that is highly relevant to landscape design and management; indeed, it could be argued that a large proportion of landscape management operations are about preventing, promoting or diverting succession (although they are not often described in such terms). Succession differs from the cyclical changes and fluctuations described above in that it involves directional change in vegetation. Whilst cycles and fluctuations imply some sort of change within a defined vegetation type (i. e. although the precise species composition may change within a grassland, the vegetation remains as grassland), successional change implies a change not only in species composition, but also in vegetation character (i. e. grassland changes to woodland) (Figure 4.9).
In landscape terms, succession can be regarded as a force, constantly driving vegetation to alter its state, both in character but also species composition from the initial starting point. But just what is the end point? Classic succession theory suggests that in any given site, the progress of successional change can be predicted to a more or less predetermined outcome or ‘climax’ vegetation that is suited to a particular climate zone, with increasing species diversity, and structural complexity of vegetation as succession proceeds. However, this concept has been largely discredited and most ecologists recognise that ‘mature’ vegetation is far more dynamic, with many different mature species assemblages occurring in any climatic region, and that the mature vegetation is in a constant state of flux, subject to cycles and fluctuations caused by external disturbances (Burrows 1990). For our purposes, a far better model for succession is to consider that vegetation reaches an equilibrium over time with the balance of environmental stress and disturbance factors that are operating on that site. The further away from that equilibrium that the vegetation is, the greater is the energy input required to keep it in that state. For example, maintaining short amenity grassland in lowland England requires far greater inputs of energy than maintaining deciduous woodland. This model has important implications because it suggests that the outcome of succession can be manipulated by altering the intensity of stress and disturbance operating on the system. It also suggests that succession itself can be used to guide vegetation to a state that is ‘sustainable’ (i. e. on any given site it can be maintained with minimal resource inputs).
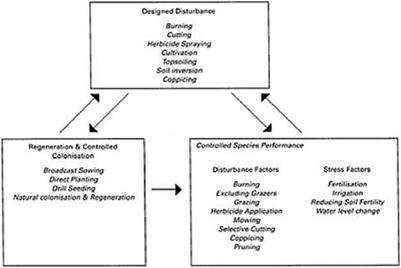
Although succession has been researched and discussed in depth for many decades, the application of this knowledge has been limited in natural conservation management (Luken 1990), and has certainly been rarely discussed in urban amenity landscape management. However, for ecologically-informed landscape vegetation, a succession – based approach provides a rational basis for creative and informed management. Luken (1990), summarising succession-based management models developed in the context of restoration ecology, proposed three main components of succession management: designed disturbance, controlled colonisation and controlled species performance. These terms are equally applicable to the management of naturalistic landscape plantings.
Designed disturbance refers to those factors that initiate new successions and vegetation
|
|
4.10
A succession-based model for the management of designed urban vegetation (arrows indicate sequential operations). Adapted from Luken, 1990
development, set back or slow down succession, or maintain cyclical change, i. e. periodic rejuvenation. In essence, designed disturbance is an artificial or human-induced operation that promotes suitable conditions for the establishment of new species or individuals on to a site. In most instances this will involve the removal of competition from existing vegetation and probably the creation of patches or areas of bare ground for seeding or planting. Some operations that can be used to create designed disturbance are listed in Figure 4.10. In effect, designed disturbances create or eliminate sites where succession can be initiated.
Controlled colonisation involves the manipulation of plant species’ availability and establishment. Figure 4.10 lists operations that either directly introduce propagules of desired species, or selectively encourage certain species to establish or regenerate from species and propagule pools already present. It is clear that where nonnative or ornamental species are to be included in a planting scheme, then artificial introduction is necessary. But even where vegetation is to be based on common native species, natural colonisation on its own is rarely satisfactory, partly because of the timescales involved and also because the resultant vegetation is likely to be composed predominantly of weedy ruderal species, at least in the short to medium term. Controlled colonisation and establishment increases or decreases the availability of plant species, according to whether they are desirable or not.
Controlled species performance includes techniques that increase or decrease the growth and reproduction of plant species to shape both the composition and form of the vegetation over time. We can relate the operations listed in Figure 4.10 that differentially control species performance to Plant Strategy Theory, discussed earlier in this chapter. Some of these factors, such as increasing or reducing soil fertility, or controlling water availability, differentially affect the rate of growth of species and can be termed stress factors, whilst others, such as grazing, mowing and pruning, selectively remove or damage plant biomass and, therefore, can be termed disturbance factors.
Figure 4.10 illustrates these three components as an integrated model for the management of landscape and garden plantings. Examples of applications of this model are given in James Hitchmough’s chapter on herbaceous plantings (Chapter 6) and also more specifically in Hein Koningen’s chapter on the creative management of ecological plantings (Chapter 10). The arrows in Figure 4.10 indicate direct sequential steps. Each component may also be repeated through time (for example, regular coppicing of woody plants or annual hay cutting of perennial meadows).
References
—Burrows, C. J. (1990). Processes of Vegetation Change. Unwin Hyman, London.
—Dickinson, G. and Murphy, K. (1998). Ecosystems. Routledge, London.
—Dunnett, N. (1995). Harnessing Anarchy. Landscape Design, Nov., 25-29.
—Dunnett, N. and Willis, A. (2000). The dynamics of Chamanerion angustifolium over a 38-year period in road verges at Bibury, Gloucestershire. Plant Ecology, 148, 43-50.
—Greig-Smith, P. (1964). Quantitative Plant Ecology. Butterworths, London.
—Grime, J. P. (1987). Dominant and subordinate components of plant communities: implications for succession, stability and diversity. In Gray, A. J., Crawley, M. J. and Edwards, P. J. (eds) Colonisation, Succession and Stability. Blackwell Scientific Publications, Oxford, pp. 413-428.
—Hansen, R. and Stahl, F. (1993). Perennials and their garden habitats. Cambridge University Press, Cambridge.
—Hitchmough, J. D. (1994). Natural Neighbours. Landscape Design, 229, 16-24.
—Kaplan, R. and Kaplan, S. (1989). The Experience of Nature. Cambridge University Press, Cambridge.
—Knops, J. M.H. etal. (1999). Effects of plant species richness on invasion dynamics, disease outbreaks, insect abundance and diversity. Ecological Letters, 2, 286-294.
—Luken, J. (1990). Directing Ecological Succession. Chapman & Hall, London.
—McCann, K. S. (2000). The diversity and stability of ecosystems. Nature, 405, 228-233.
—Pielou, E. C. (1961). Segregation and symmetry in two-species populations as studied by nearest neighbour relationships. Journal of Ecology, 49, 255-269.
—Purvis, A. and Hector, A. (2000). Getting the measure of biodiversity. Nature, 405, 212-219.
—Schwartz, M. W. et al. (2000). Linking biodiversity to ecosystem functioning: implications for conservation ecology. Oecologia, 122, 297-305.
—Tilman, D., Knops, J., Wedin, D., Reich, P., Ritchie, M. and Siemann, E. (1997). The influence
of functional diversity and composition on ecosystem processes. Science, 277, 1300-1302.
—Watt, A. S. (1947). Pattern and process in the plant community. Journal of Ecology, 35, 1-22.
—Watt, A. S. (1971). Factors controlling the floristic composition of some plant communities in Breckland. In The scientific management of animal and plant communities for conservation. Blackwell Scientific Publications, Oxford.