Fertilizers are nutrient additives applied to the soil periodically to maintain optimum crop productivity. The need for fertilization may result from a deficiency of one or more mineral elements in the soil, their presence in a form unavailable to the plant, or the leaching of elements into the soil to a depth below the root zone.
Chemical Fertilizers
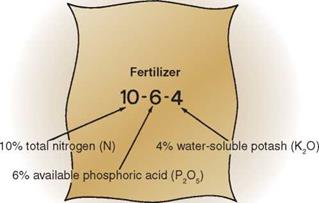
Since nitrogen, phosphorus, and potassium are the soil elements used in greatest quantity by the green plant, a fertilizer that provides all three elements is termed a complete fertilizer. The actual percentage by weight of each of the three primary elements in a fertilizer determines its analysis; for example, 100 pounds of 10-6-4 analysis fertilizer contains 10 pounds of nitrogen (N), 6 pounds of phosphoric acid (P2O5), and 4 pounds of potash (K2O). The analysis figures are always expressed in the same order and represent the same nutrients. Note that phosphorus and potassium are not present in elemental form, but as chemical compounds. Thus the amount of actual element provided for the plant is less than the analysis implies (Figure 3-7).
With simple arithmetic, fertilizers can be compared on the basis of their nutrient ratio. The ratio is a reduction of the analysis to the lowest common denominator. For example, a 5-10-10 analysis has a ratio of 1-2-2. (Each of the numbers has been reduced through dividing by a common factor of 5.) A fertilizer analysis of 10-20-20 also has a ratio of 1-2-2. Therefore, a 5-10-10 fertilizer supplies the three major nutrients in the same proportion as a 10-20-20 fertilizer, but twice as much of the actual product must be applied to obtain the same amount of nutrients (Table 3-3).
When less than 30 percent of a complete fertilizer’s weight represents available nutrients, it is termed a low-analysis fertilizer. When the amount of available nutrients is 30 percent or more, the product is a high-analysis fertilizer. The remaining material in a fertilizer is filler, either organic or synthetic. The filler may provide some additional essential elements, and may even be important as a source of micronu-
|
|
figure 3-7. Interpreting fertilizer analysis figures: the nutrients are always shown in the same order. (Delmar/ Cengage Learning)
|
TABLE 3-3.
|
trients, but essentially the filler is a carrier for the available macronutrients. It allows them to be applied evenly to the soil and crop. It also adds weight and bulk, both undesirable features. Table 3-4 compares high-analysis and low-analysis fertilizers.
The more common chemical fertilizers include: anhydrous ammonia, ammonium nitrate, urea, sodium nitrate, and ammonium sulfate as carriers of nitrogen; superphosphates, ammoniated phosphates, and round rock phosphate as carriers of phosphorus; and potassium nitrate, potassium chloride, potassium sulfate, and potassium-magnesium sulfate as carriers of potassium.