Metal is used in both the form and structure of furniture as well as for hardware and mechanical fasteners. It is, however, a material considered uncomfortably cool to the touch
and therefore is conventionally used for supportive elements in furniture. It is a remarkable material that can be curved, bent, and rolled. Stainless steel, aluminum, brass, and bronze are commonly used metals in contemporary furniture (Figure 7.39). Ludwig Mies van der Rohe and Poul Kjsrholm were both renowned for their preference for using stainless steel rather than chrome-plated steel in most of their furniture.

 Metals are consistent in quality, stability, and hardness. Metal is a homogeneous material (without grain) and is given texture through a variety of techniques. It can be worked to precise dimensions far greater than those of wood, glass, or stone. Metal can be joined by being screwed, bolted, or welded. When joining different metals, keep in mind that galvanic action will occur when dissimilar metals touch and are exposed to moisture. Galvanic action is a chemical reaction that occurs in the presence of humidity or moisture and will corrode one or the other metal when two metals are in contact.
Metals are consistent in quality, stability, and hardness. Metal is a homogeneous material (without grain) and is given texture through a variety of techniques. It can be worked to precise dimensions far greater than those of wood, glass, or stone. Metal can be joined by being screwed, bolted, or welded. When joining different metals, keep in mind that galvanic action will occur when dissimilar metals touch and are exposed to moisture. Galvanic action is a chemical reaction that occurs in the presence of humidity or moisture and will corrode one or the other metal when two metals are in contact.
Aluminum is a relatively soft, lightweight metal that can be cast in sand, rolled, extruded, forged, and drawn (Figure 7.40). However, it is very difficult to weld. It is an exceptional material for outdoor use because its outermost layer converts to aluminum oxide when exposed to air, giving aluminum a nearly transparent layer that resists further corrosion. Pure aluminum has excellent electrical and thermal conductivity (as compared to copper). In the mid – 1800s, aluminum was more expensive than silver. Thanks to an abundance of aluminum-bearing ores (bauxite) and development of modern means of extracting the mineral, aluminum today is widely available and inexpensive. It is the third most abundant metal in the earth’s crust (after iron and silicone).
Brass is an alloy of copper and zinc that produces a soft yellow metal. Typically, brass is two-thirds copper and one-third zinc. It can be filed, sawn, drilled, bent, and braised. Brass is used as a base metal for plating other metals and finishes, making it an excellent choice for architectural or cabinet furniture hardware. Secondary uses include screws and decorative work.
Bronze was the first metal used in furniture. It is an alloy of copper and tin and is an excellent material for locks, hinges, and decorative elements because it is resistant to corrosion.
Copper saws, files, and solders easily. Though it polishes easily, copper will patina to a brown and then to a green color (copper carbonate) in clean air, and to a black
color (copper sulfide) in dirty air. Copper and its alloys are easy to cast, to roll to sheet, to draw to wire, and to shape in other ways. Copper and its alloys are particularly desirable for recycling.
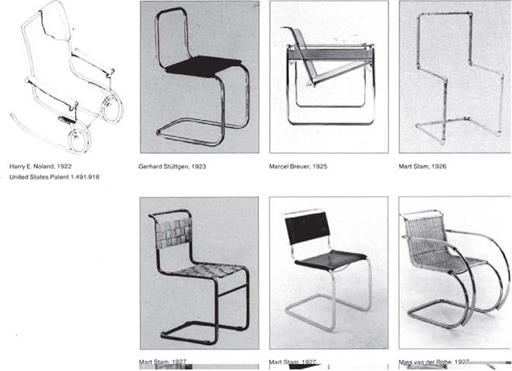
Carbon steel is an alloy of iron and has up to 1.7 percent carbon. Early-twentieth – century furniture used tubular steel bent into various shapes. Mart Stam and Marcel Breuer of the Bauhaus were among the first designers who used precision steel tubing in their furniture (Figure 7.41). Many furniture designers, beginning as early as the second half of the nineteenth century, used wire and steel rod. In the twentieth century, Harry Bertoia designed a series of wire frame chairs that are still in production (see Chapter 4). Carbon steels are easy to recycle, and little energy is needed to recycle them.
Ferrous metals are metals that have some percentage of iron in their content. The term ferrous is derived from the Latin word ferrum, which means "containing iron." These metals are generally used to manufacture extruded bar stock, which is the basic structural framing elements used to fabricate supports and framework in furniture. Nonferrous metals such as aluminum, brass, bronze, chromium, copper, nickel, and zinc are commonly used as metal finishes. Steel is often galvanized with a thin coat of zinc to minimize oxidation, but it is difficult to work. The process of galvanizing steel with zinc makes it problematic to weld or cut safely in a shop because cutting or welding galvanized steel produces a toxic off-gas. Zinc, in its pure form, was used to clad bar counters throughout France because of zinc’s properties. It does not react to acids (wine) or cleaning fluids and is easy to shape.
Although created earlier in the twentieth century by a Frenchman and a German, stainless steel was rediscovered in Sheffield, England, in 1913. Harry Brearley has been credited with popularizing the material. Made of iron with about 13 percent chromium and a small portion of carbon, stainless steel will not rust, remains ductile even at low temperatures, and welds easily. It is an excellent material for use in food service. There are many alloys of stainless steel,
![]()
|

 which include USD316, USD321, USD306, and USD630. USD304 is the typical grade, composed of 74 percent iron, 18 percent chromium, and 8 percent nickel. Stainless steel is selected mainly for its resistance to corrosion, its mechanical resistance, and its workability.
which include USD316, USD321, USD306, and USD630. USD304 is the typical grade, composed of 74 percent iron, 18 percent chromium, and 8 percent nickel. Stainless steel is selected mainly for its resistance to corrosion, its mechanical resistance, and its workability.



